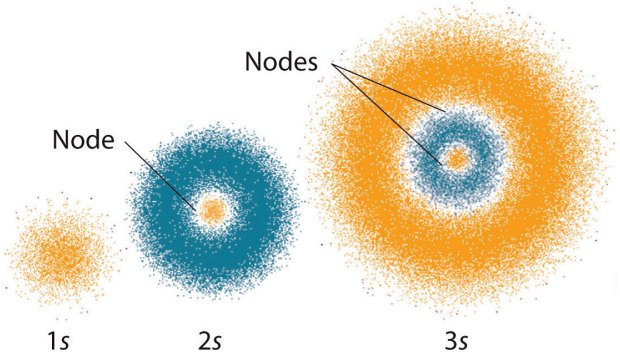
Take the derivative with respect to #r#.#r#, one thing we can do is find the region of highest electron density, which is just the highest point in the #1s# orbital graph above. Now, since we have the plot for #4pir^2R_(nl)^2(r)# vs. This is the function that corresponds to the red curve in the diagram above. In order to plot the "probability pattern", we have to grab what we need and turn it into #4pir^2(R_(nl)(r))^2#. The simplest example of an orbital wave function (used by #"Real Chemists"^"TM"#, folks!) is the one for the #\mathbf(1s)# orbital, which happens to be: This will give a result that should prove familiar. The wave functions themselves, as we defined them, generate these "probability patterns". When we plot the radial density distribution (or "probability pattern") of an orbital, we plot #4pir^2(R_(nl)(r))^2# vs. OKAY, WHAT DOES IT HAVE TO DO WITH PROBABILITY PATTERNS?įocus on the #R_(nl)(r)# portion of #psi#. It depends on the quantum numbers #\mathbf(l)# and #\mathbf(m_l)#. #Y_(l)^(m_l)# is the angular component of the wave function, describing the aspects of the orbital electron distribution that can possibly give it a non-spherical shape.It depends on the quantum numbers #\mathbf(n)# and #\mathbf(l)#. 
#R_(nl)# is the radial component of the wave function, describing the variation in the distance from the center of the orbital (the radius!).It depends on the quantum numbers #\mathbf(n)#, #\mathbf(l)#, and #\mathbf(m_l)#. #psi_(nlm_l)# is a wave function that can be constructed to describe what the orbital's electron distribution looks like.#r#, #theta#, and #phi# are spherical coordinates:.In general, the wave function for spherical harmonics coordinates can be written as: WHAT IS THE WAVE FUNCTION FOR AN ORBITAL?Įrwin Schrodinger published the wave function #psi#, which describes the state of a quantum mechanical system.įor each orbital, its radial density distribution describes the regions with particular probabilities for finding an electron in that particular orbital. You can imagine anything you want for what "determines" the probability distribution, but ultimately, nature turned out that way, and we described these orbitals after-the-fact.ĭISCLAIMER: This is going to be a long answer. We can't define equations to influence nature, only describe it.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
